Light Activation
Most molecules absorb light, and in so doing they absorb energy that promotes an electron to an excited state. The energy from this excited state can lead to a number of useful processes. The prime example of this type of chemistry in nature is the action of chlorophyll in plants; it absorbs light and transfers energy through the protein photosystems to produce chemical energy.
Most molecules absorb light, and in so doing they absorb energy that promotes an electron to an excited state. The energy from this excited state can lead to a number of useful processes. The prime example of this type of chemistry in nature is the action of chlorophyll in plants; it absorbs light and transfers energy through the protein photosystems to produce chemical energy.
|
Pyrrolic Macrocycles
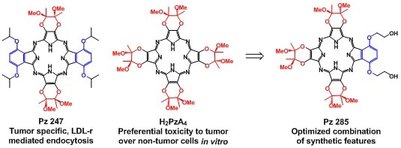
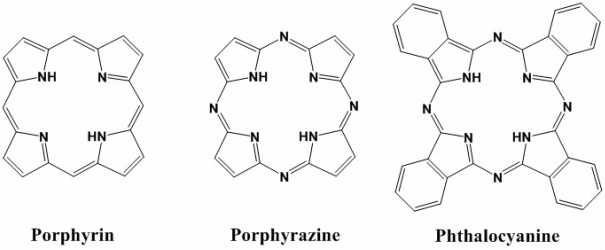
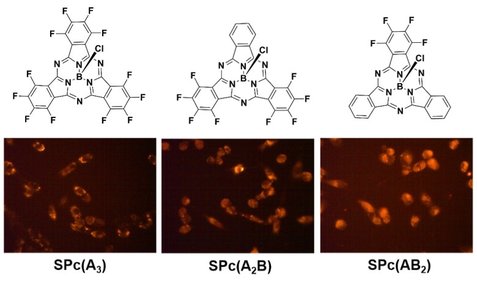
Tetrapyrroles are abundant in nature. They are what make plants green and our blood red. These brilliant colors are indicative of a tetrapyrrole's ability to absorb light, thereby being activated for some useful function. They are fluorescent and can react with molecular oxygen to produce toxic reactive oxygen species (ROS). They are rigid and planar, allowing them to bind DNA, and hydrophobic, promoting interactions with blood proteins. These properties, taken together, make tetrapyrrolic macrocycles excellent candidates as diagnostic/therapeutic agents for a range of diseases, including cancer and myocardial infarction. We intend to develop synthetic methods for the high-yield production of low symmetry tetrapyrroles. |
|
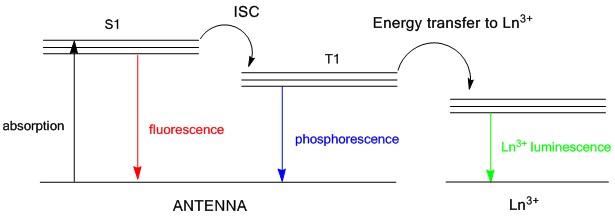
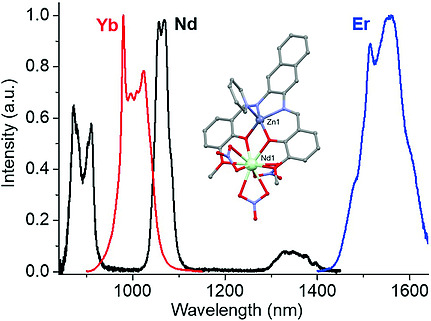
Lanthanide Coordination Chemistry
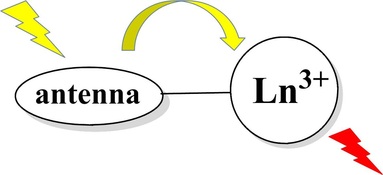
Lanthanide metals are of the rare-earth family but they are not all that rare. Members of the lanthanide series are abundant on earth at the same order of magnitude as nickel, copper, and zinc. Furthermore, lanthanides possess the electronically unique f-orbitals that impart many interesting properties. We are particularly interested in the metal-based luminescence of lanthanide ions. Sensitization of this luminescence requires an organic "antenna" to transfer energy and we aim to develop proper ligand systems to acheive this task. An important parameter that we hope to exploit is a ligand system with synthetic tunability of electronic structure, while allowing the selective addition of solubleizing groups for application in biological systems. |